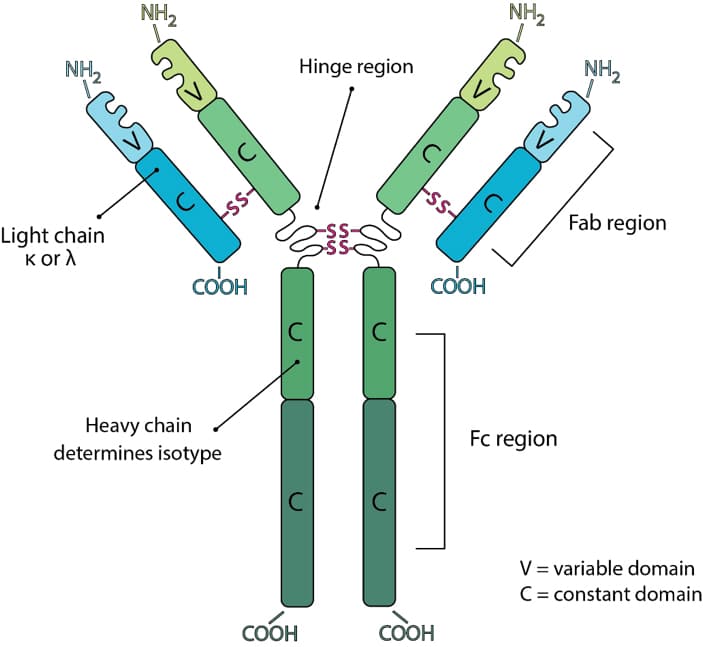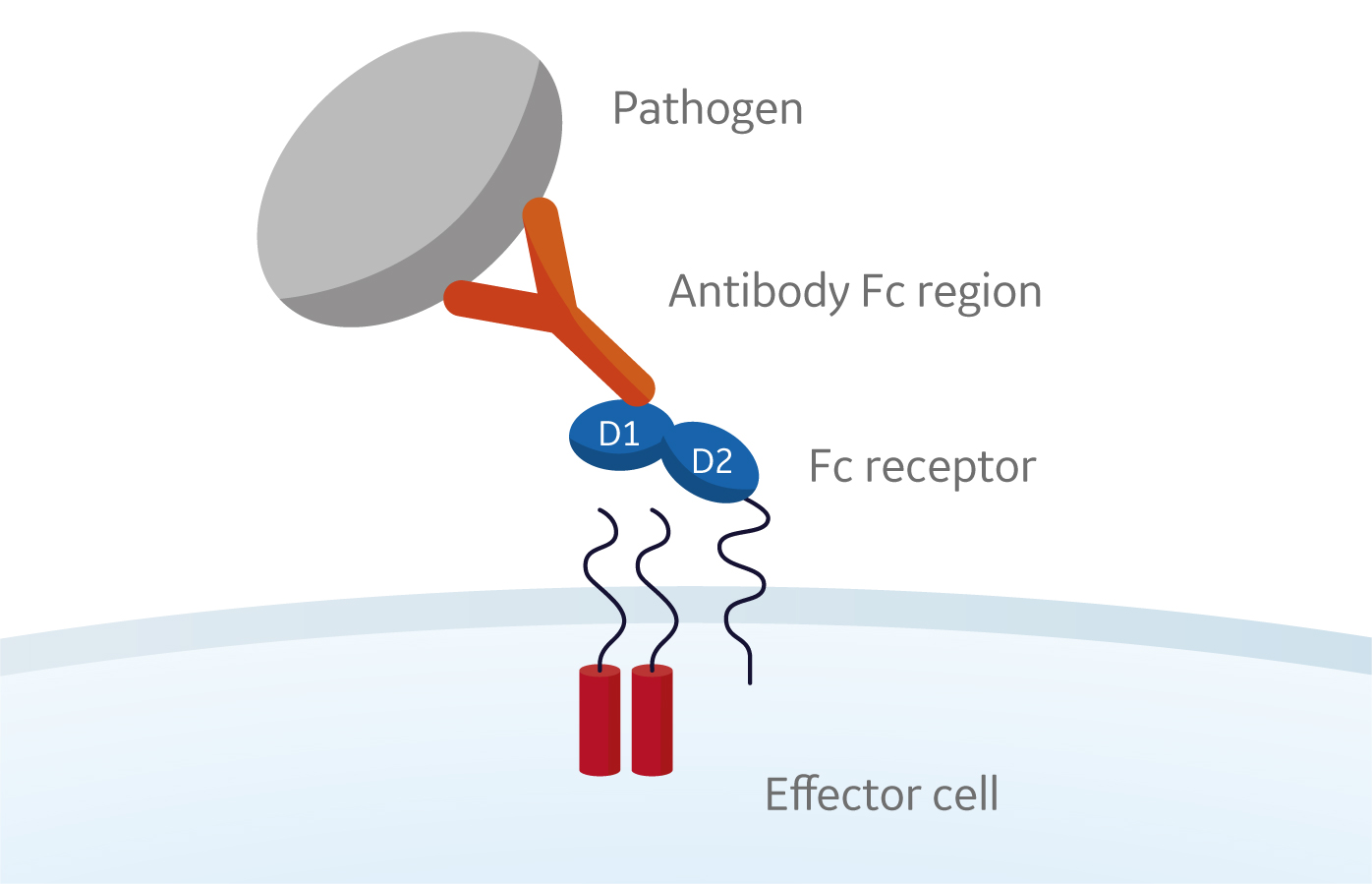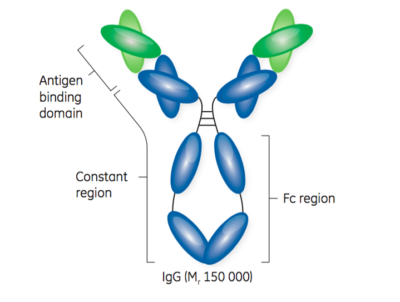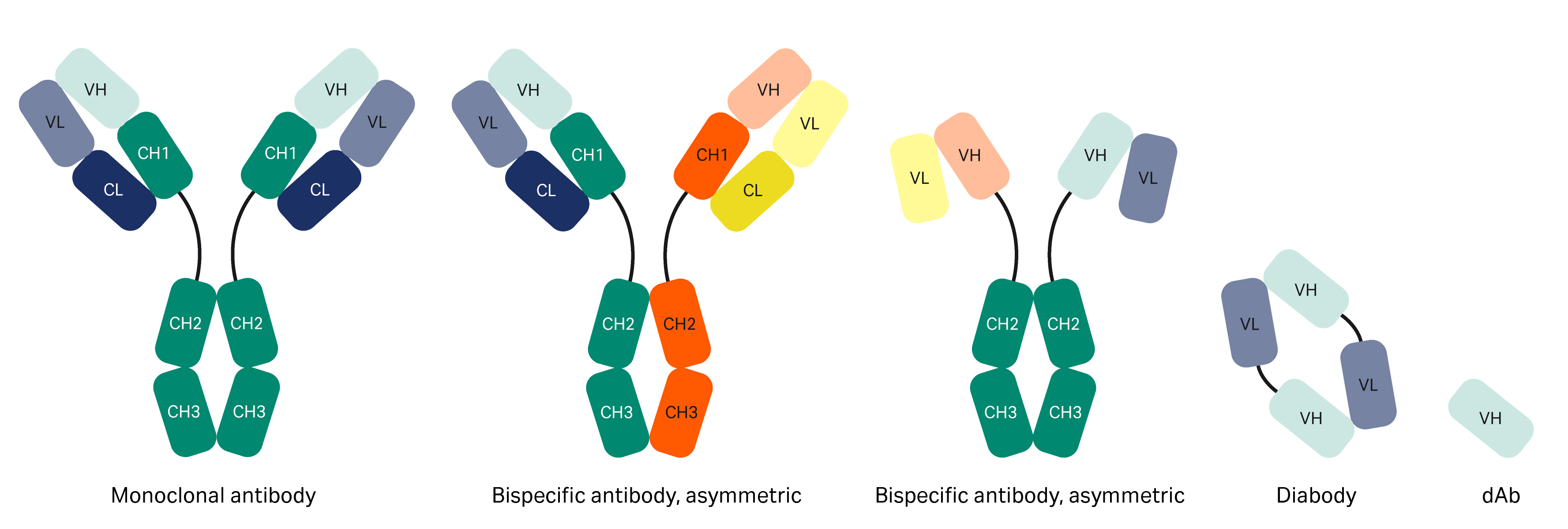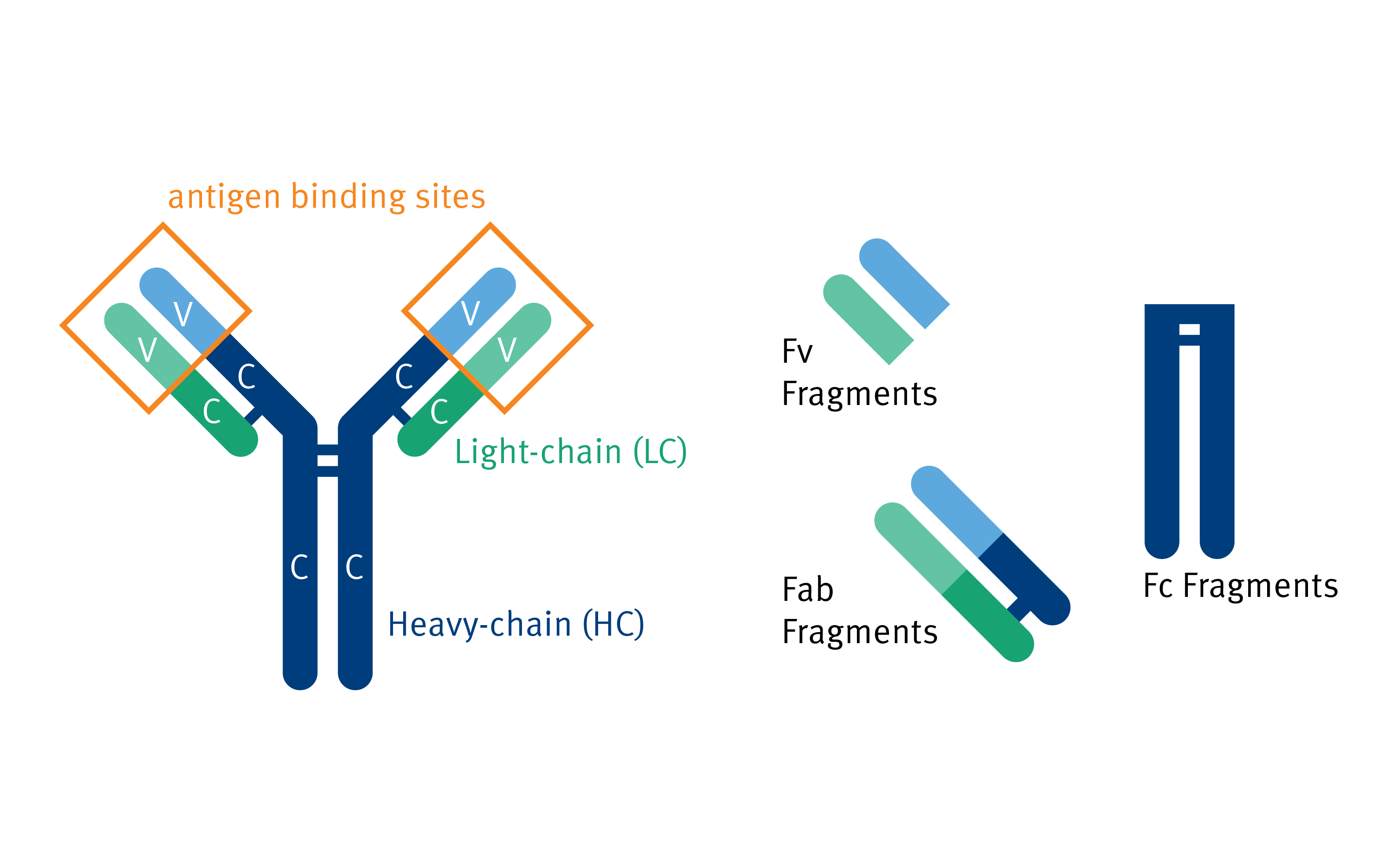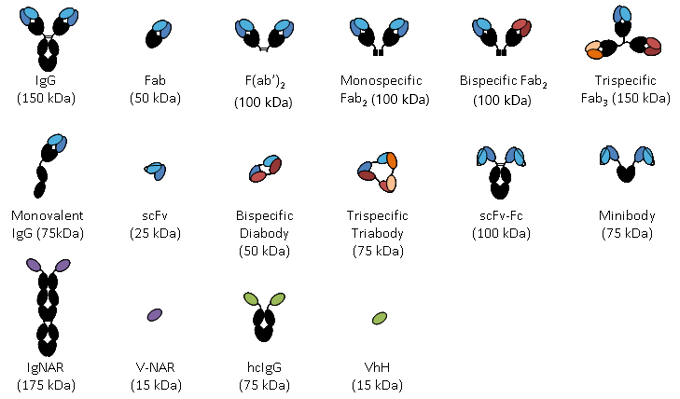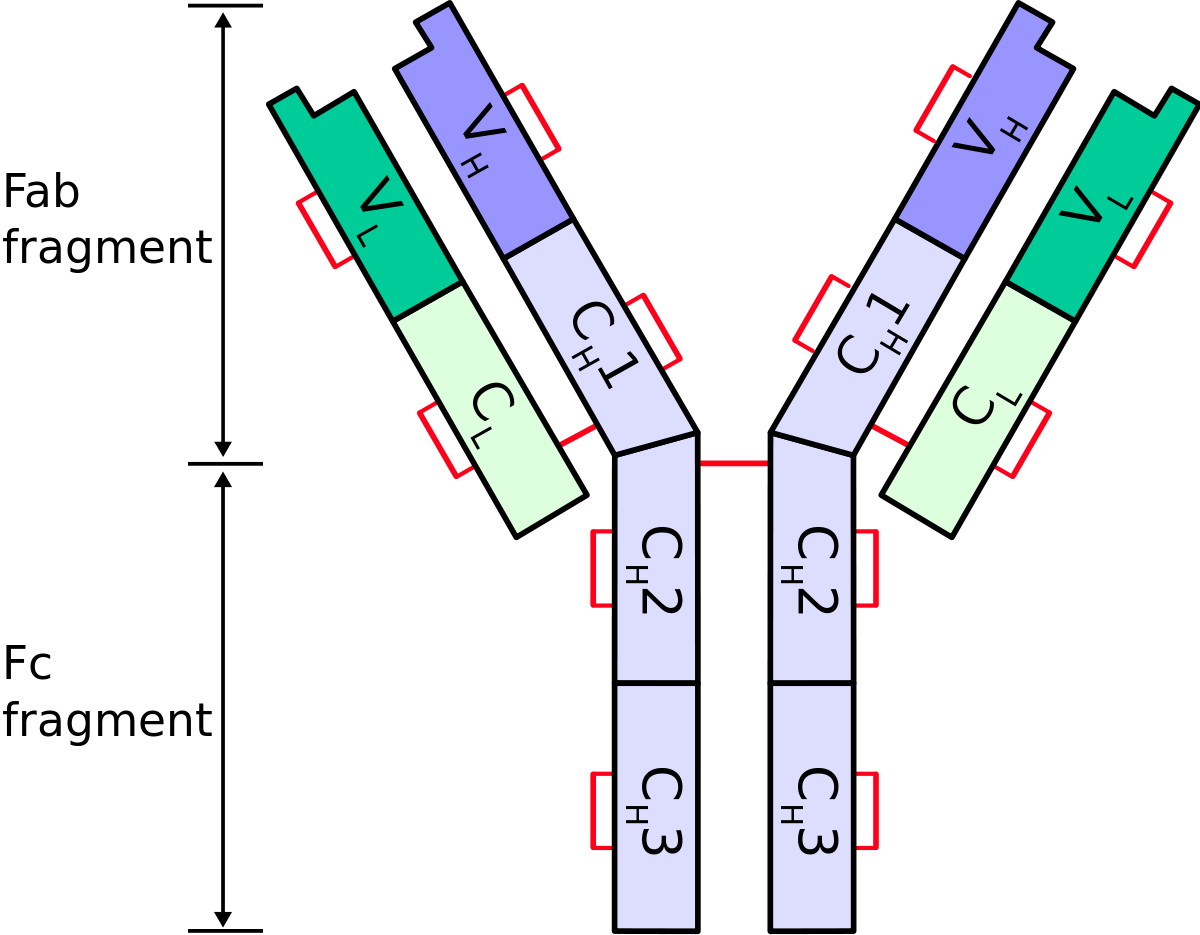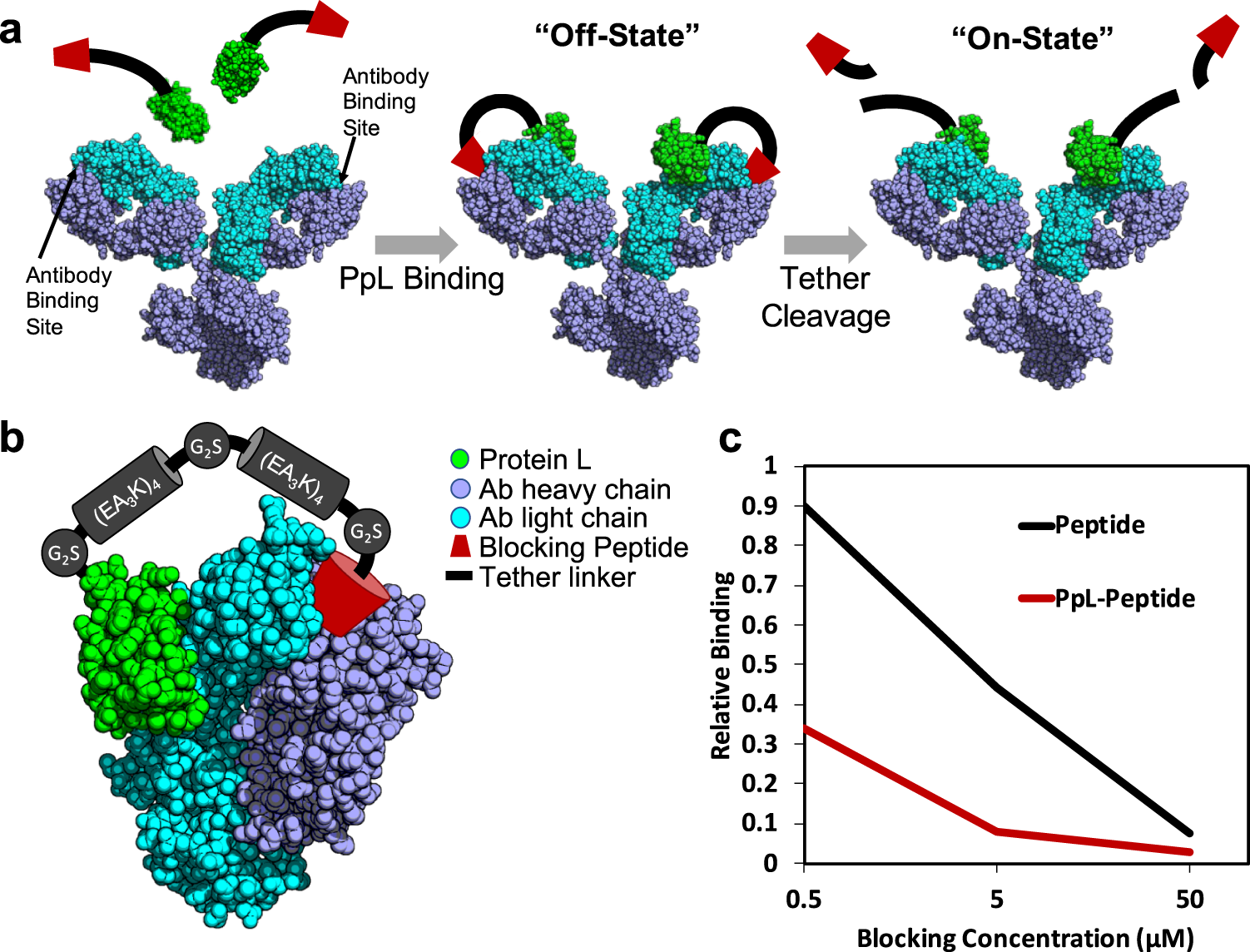
Turning antibodies off and on again using a covalently tethered blocking peptide | Communications Biology
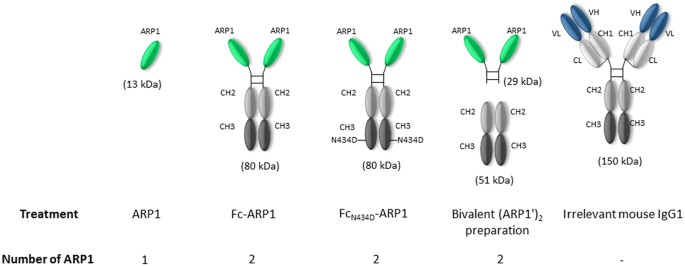
Fusion of the mouse IgG1 Fc domain to the VHH fragment (ARP1) enhances protection in a mouse model of rotavirus | Scientific Reports
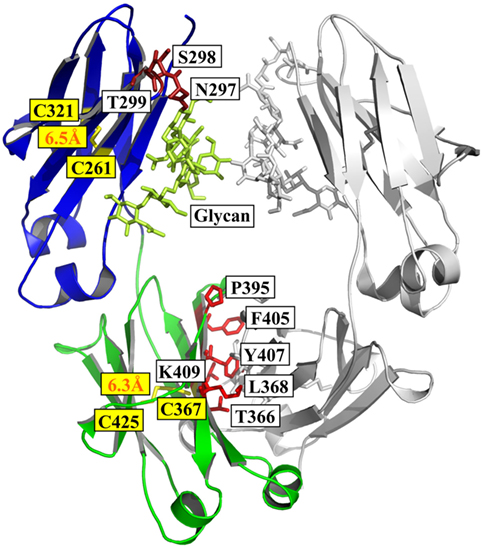
Frontiers | Engineering of Fc Fragments with Optimized Physicochemical Properties Implying Improvement of Clinical Potentials for Fc-Based Therapeutics
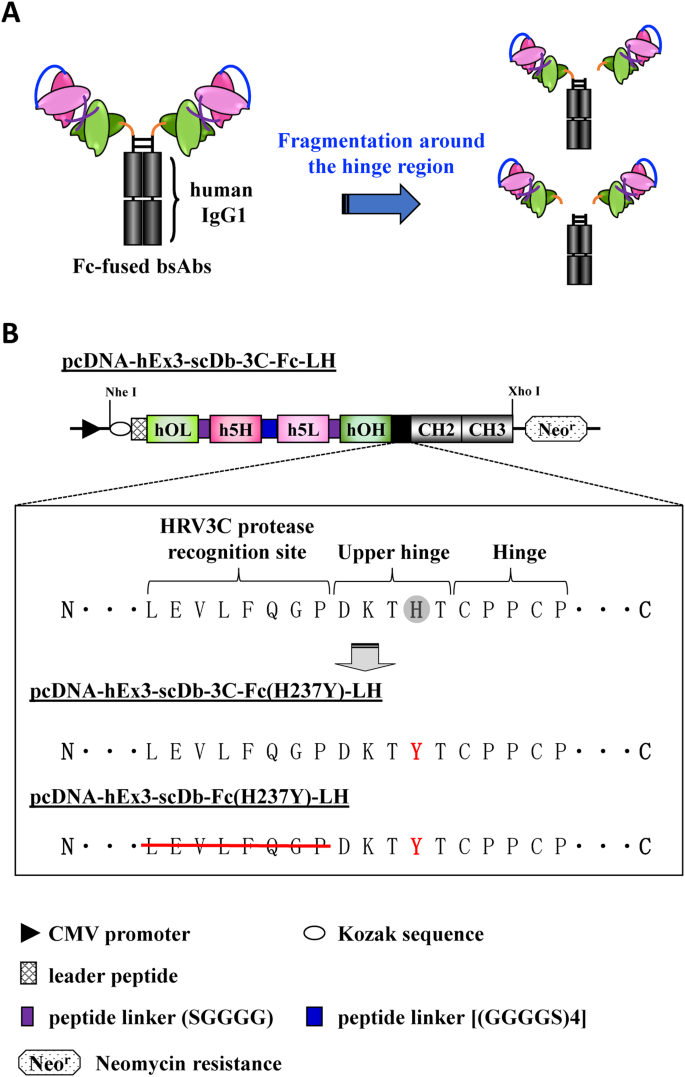
Engineering the hinge region of human IgG1 Fc-fused bispecific antibodies to improve fragmentation resistance | Scientific Reports